by H.S. Sterling, Jul 5, 2025 in WUWT
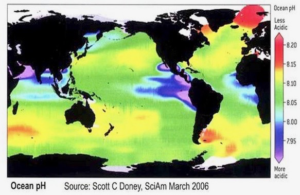
The U.K.’s The Guardian ran an article claiming that the world’s oceans have surpassed a critical tipping point in acidity threatening sea life. This is false. The pH content of the world’s oceans varies by time and place throughout the day, rising and falling modestly, but the average pH content remains far from acidic and there is no evidence crustaceans or other types of shellfish are being threatened by the sea water becoming acidic.
Lisa Bachelor, the writer of The Guardian’s article, “‘Ticking timebomb’: sea acidity has reached critical levels, threatening entire ecosystems – study,” says:
The world’s oceans are in worse health than realized, scientists have said today, as they warn that a key measurement shows we are “running out of time” to protect marine ecosystems.
Ocean acidification, often called the “evil twin” of the climate crisis, is caused when carbon dioxide is rapidly absorbed by the ocean, where it reacts with water molecules leading to a fall in the pH level of the seawater. It damages coral reefs and other ocean habitats and, in extreme cases, can dissolve the shells of marine creatures.
Bachelor’s story is based upon a study which claims that ocean acidity has breached a “planetary boundary,” the seventh of nine such milestones or boundaries to be breached, threatening to cause permanent damage to the planet’s health.
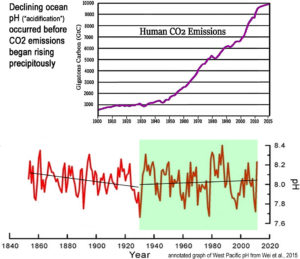
The study looked at ice core records and studies of marine life, run through algorithms of complex computer models to assess the past 150 years, concluding the ocean acidification boundary had been breached, with the world facing a “ticking timebomb,” of sea life destruction.
This study’s findings are driven by woefully flawed computer models, a limited time horizon and understanding of long-term history, and lack a basis in real world data. As such it and The Guardian’s dire warnings based on it, are unjustified.
Model outputs are only as good as the assumptions, data, and our understandings of the feedbacks and systems built into them. Even as our knowledge improves, our understanding of the oceans and the interactions of its various currents, systems, inputs, and outputs remain limited, thus the assumptions built into the models are weak and uncertain. As discussed at Climate Realism, here, here, and here, for example, the climate model outputs fail to match reality.

 I
I